Unlike the competition, RemeGenix' lead drug in its pipeline is a
Modulator
of Beta secretase processing of APP (MoBA)
and, as such, can reduce APP processing without having the potential for side effects that have been
seen when using various secretase inhibitors (see
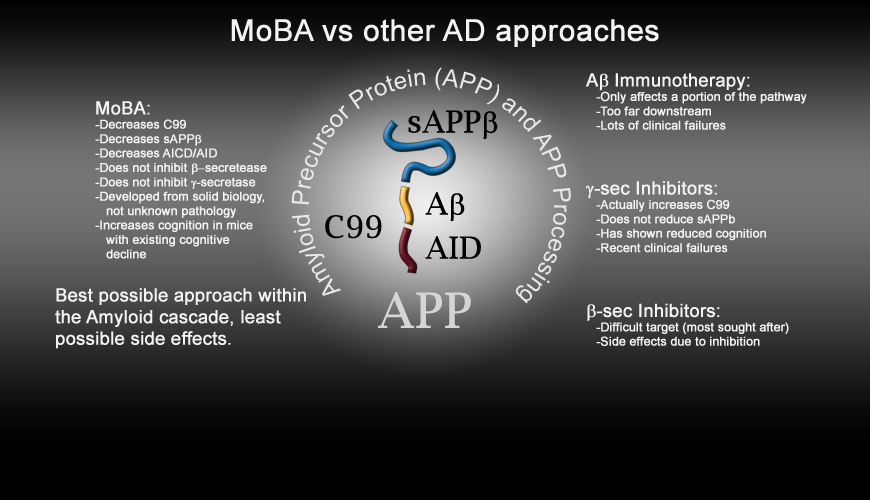
here for an overview of the activities of MoBA in comparison with the competitive products).
Thus, MoBA provides the most specific protection, and therapeutic recovery from the negative biological processes that cause
cognitive dysfunction.
In early proof of concept animal studies, the company's scientific founder, Luciano D’Adamio, at the Albert Einstein College of Medicine, demonstrated that the therapeutic approach (of which MoBA is now the lead candidate) can act to alter the progression of the disease, possibly even after manifestation of a memory deficit, and MoBA has been shown to directly modulate APP processing. Taken together, RemeGenix' lead drug candidates represent a novel disease altering mechanism, and have the potential to treat both early (asymptomatic) and late stage (symptomatic) disease.
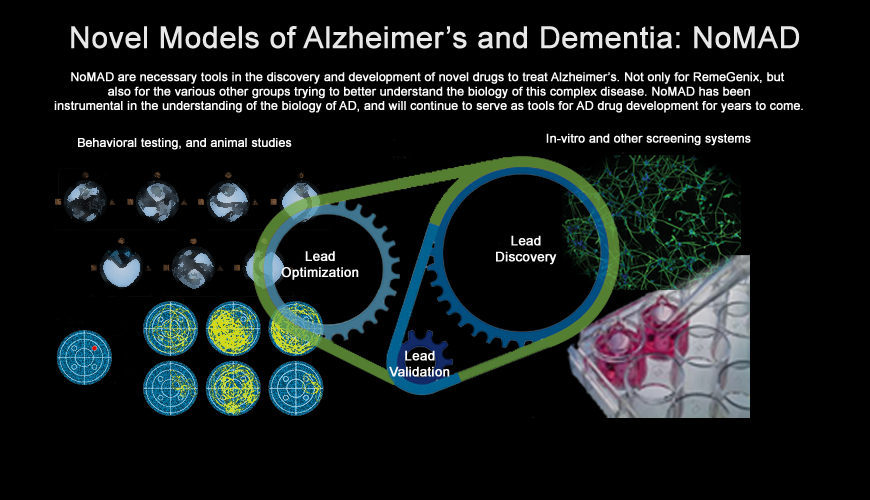
In discovery efforts, unsatisfied with existing animal models for dementia, Dr. D’Adamio created our Novel animal Models of Alzheimer's and dementia (NoMAD), and then used these models to advance our scientific understanding of the disease. NoMAD was also used to establish proof of principle of MoBA in direct comparison with several competitive drugs acting in the APP pathway. NoMAD is now patent pending and licensed to RemeGenix, and represents the first biological model of AD that faithfully replicates a human dementia. We anticipate, as more and more people review NoMAD, and as the industry's understanding of the biology of AD increases, that they will realize the need for such a model in their own discovery efforts (see here for an overview of the type of discovery tools possible with NoMAD).
The Company feels strongly, and has openly stated, that many of the competitive products in development have failed clinical trials because they were designed and tested on animal models that are not biologically relevant, meaning that they do not genetically match any possible form of human dementia, and thus the results achieved with models other than NoMAD are almost guaranteed to fail once translated into human studies.
In early proof of concept animal studies, the company's scientific founder, Luciano D’Adamio, at the Albert Einstein College of Medicine, demonstrated that the therapeutic approach (of which MoBA is now the lead candidate) can act to alter the progression of the disease, possibly even after manifestation of a memory deficit, and MoBA has been shown to directly modulate APP processing. Taken together, RemeGenix' lead drug candidates represent a novel disease altering mechanism, and have the potential to treat both early (asymptomatic) and late stage (symptomatic) disease.
In discovery efforts, unsatisfied with existing animal models for dementia, Dr. D’Adamio created our Novel animal Models of Alzheimer's and dementia (NoMAD), and then used these models to advance our scientific understanding of the disease. NoMAD was also used to establish proof of principle of MoBA in direct comparison with several competitive drugs acting in the APP pathway. NoMAD is now patent pending and licensed to RemeGenix, and represents the first biological model of AD that faithfully replicates a human dementia. We anticipate, as more and more people review NoMAD, and as the industry's understanding of the biology of AD increases, that they will realize the need for such a model in their own discovery efforts (see here for an overview of the type of discovery tools possible with NoMAD).
The Company feels strongly, and has openly stated, that many of the competitive products in development have failed clinical trials because they were designed and tested on animal models that are not biologically relevant, meaning that they do not genetically match any possible form of human dementia, and thus the results achieved with models other than NoMAD are almost guaranteed to fail once translated into human studies.
| Pipeline ID | Research & Development | Lead Optimization | Clinical Development | Commercialization |
| MoBA |
|
|||
| NoMAD |
|
|||
|
RemeGenix' scientific founder, Dr. D'Adamio has developed an innovative and novel suite of therapeutic product candidates for use in treating
neurodegenerative diseases such as Alzheimer's and dementia.
Our lead technology, MoBA, derived from the BRI2 protein, has demonstrated commercial potential in several animal models, and has provided evidence
that MoBA can work therapeutically. |
|
Preclinical studies of MoBA and its parent protein BRI2, have demonstrated significant efficacy and safety. MoBA's specificity in binding APP provides us with hints of a promising long-term safety profile. So far, MoBA has shown the following results:
|
|
Collectively, our research efforts provide significant evidence of safety and efficacy of our product portfolio, and the Company is solidly moving forward on two primary initiatives:
|